Author: Rob Visor
Learn how the FDA uses FAERS, Sentinel, and AI to monitor generic drugs after approval, ensuring safety and bioequivalence for millions of users.
View DetailsLearn when to use OTC medications versus prescription drugs. Understand potency, FDA regulations, and the safety signs that mean you need a doctor's visit.
View DetailsDiscover the urgent steps to replace lost or stolen prescription medications while traveling internationally. Learn about insurance claims, local pharmacy laws, and safety tips.
View DetailsLearn exactly how to report a medication safety concern to your clinic, including preparing evidence, contacting the safety officer, and understanding the review process.
View DetailsLearn how to effectively report medication safety concerns to your clinic. This guide covers what details to gather, who to contact, expected timelines, and how to ensure your report leads to real safety improvements.
View DetailsLearn how to verify lot numbers and expiration dates when clearing expired medication stock. Includes step-by-step protocols, recall checklists, and FDA compliance tips.
View DetailsLearn how to track lot numbers and handle medical recalls safely. This guide explains what lot numbers are, how to find them, and actionable steps patients should take to protect their health during safety alerts.
View DetailsCost-effectiveness analysis reveals how generic drugs save billions by comparing price to health outcomes. Learn why some generics cost 15x more than equally effective alternatives-and how to spot the real savings.
View DetailsIn the U.S., only certain biosimilars are labeled 'interchangeable' - meaning pharmacists can swap them for the original drug without asking the doctor. Learn how this works, who qualifies, and why state laws make it confusing.
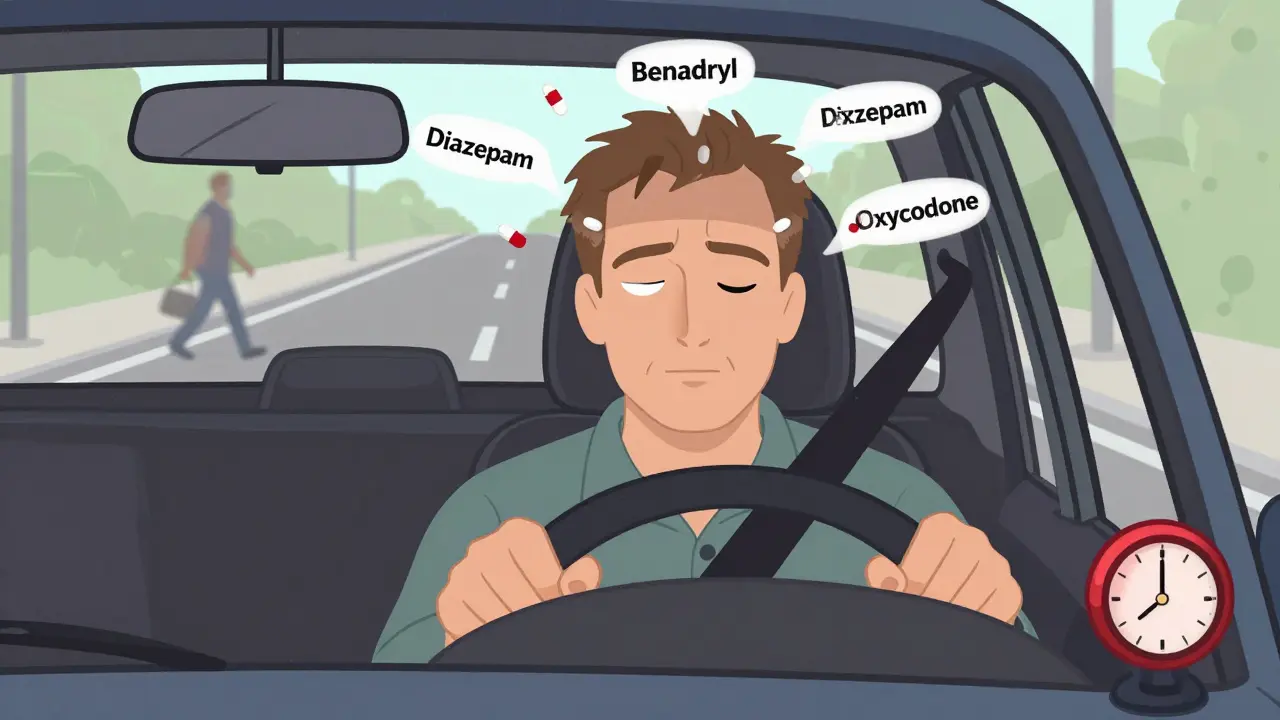
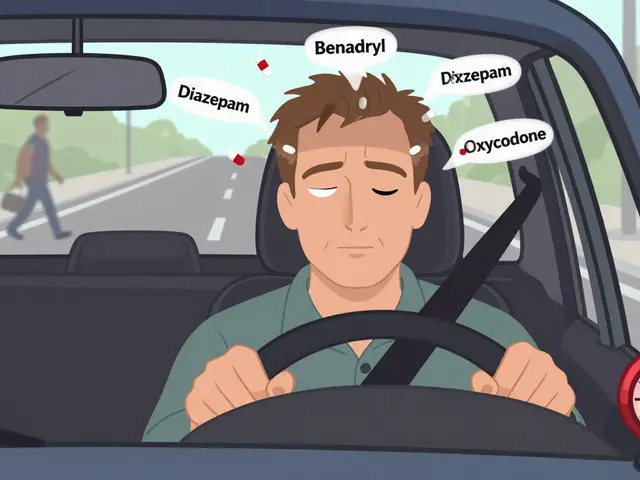
View DetailsMany common medications-prescription and over-the-counter-can severely impair driving ability, leading to crashes, legal trouble, and even death. Learn which drugs are risky, how they affect you, and what steps to take to stay safe on the road.
View Details