When you pick up a prescription for insulin or an arthritis drug, you might not realize that what the pharmacist hands you isn’t always the exact brand your doctor wrote on the script. In the U.S., some biosimilars can be swapped in automatically - no call to the doctor needed. But not all biosimilars can do this. Only those with a special label: interchangeable.
What Does "Interchangeable" Actually Mean?
Interchangeable isn’t just a fancy word for "similar." It’s a legal and regulatory status given by the FDA that lets pharmacists swap a biosimilar for the original biologic drug without asking the prescriber. Think of it like this: generic pills for high blood pressure or antibiotics have been interchangeable for decades. You get a different brand, same active ingredient, same effect. Biosimilars are different. They’re made from living cells - not chemicals - so they’re more complex. Even tiny changes in manufacturing can affect how they work in your body.The FDA created the "interchangeable" category in 2010 under the BPCIA law. Since then, only 10 out of 41 approved biosimilars have earned this status. Why so few? Because the bar is high. To be called interchangeable, a biosimilar must not only match the original in safety and effectiveness - it must also prove that switching back and forth between the two, multiple times, doesn’t cause harm or reduce results. That means clinical studies where patients get the brand, then the biosimilar, then the brand again - and nothing changes in how they feel or how their blood tests look.
Interchangeable vs. Biosimilar: The Key Difference
All FDA-approved biosimilars are safe. All are effective. That’s not up for debate. But only some are interchangeable. The difference isn’t about quality - it’s about substitution rules.A regular biosimilar needs the doctor’s OK before it’s swapped. Your doctor might choose it because it’s cheaper, but the pharmacy can’t switch it in without checking first. An interchangeable biosimilar? The pharmacist can swap it automatically - just like a generic. This matters because it changes how patients get access. In states with automatic substitution laws, a patient might never know they got a different drug unless they check the label. That’s why some people worry about being switched without warning.
Here’s the reality: the first interchangeable biosimilar was Semglee, an insulin approved in July 2021. It hit the market at 15% less than the original, and within six months, it held 17.3% of the insulin market. That’s more than double the uptake of non-interchangeable biosimilars in similar timeframes. The second big win came in August 2023 with Cyltezo, the first interchangeable biosimilar for adalimumab (Humira). That’s a huge deal - Humira was the top-selling drug in the U.S. for years. Now, patients have options.
State Laws Make It Messy
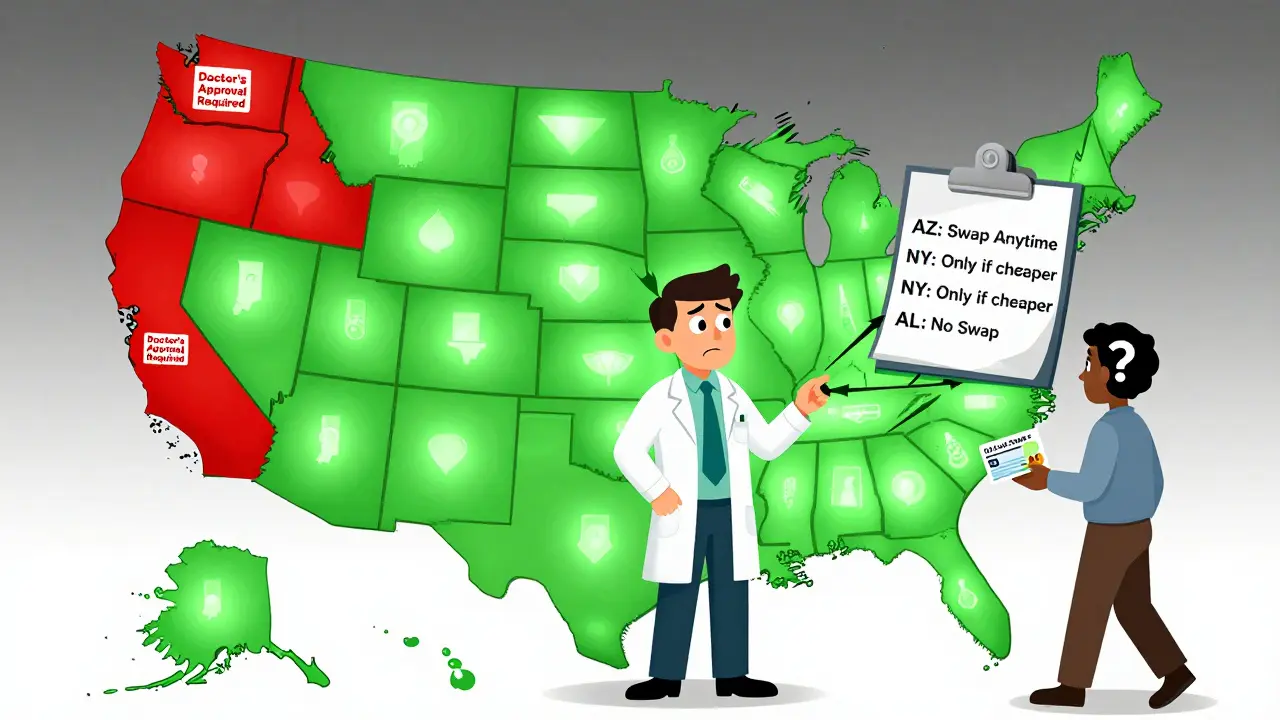
Here’s where it gets complicated. Even if the FDA says a biosimilar is interchangeable, your state might say no. As of 2023, 40 states let pharmacists substitute without asking the doctor. Arizona, California, Texas - they all allow it. But four states - Alabama, Indiana, South Carolina, and Washington - require the prescriber to give permission before any swap. Then there are six states and D.C. that only allow substitution if it saves the patient money. That means a pharmacist in New York might swap your Humira for Cyltezo, but if you live in Alabama, they can’t unless your doctor signs off.Pharmacists are stuck in the middle. A 2022 survey found that 67% of independent pharmacists said they’re confused about the rules. One pharmacist on Reddit wrote: "In California, I have to check if it’s cheaper. In Arizona, I don’t. My pharmacy software doesn’t tell me the difference." Imagine trying to fill a prescription for someone who lives in one state but gets insurance from another. The system isn’t built for that.
What Patients Experience
Patients have mixed stories. One person with psoriasis told a support forum they saved $800 a month after switching from Humira to Hyrimoz - no side effects, same results. Another said they were switched to Hadlima without warning, had an allergic reaction, and later found out it was due to a different excipient (a non-active ingredient). That’s the thing about biosimilars: while the main molecule is nearly identical, the "fillers" or stabilizers might differ. For most people, it doesn’t matter. For a few, it does.A 2022 survey from the National Psoriasis Foundation found 63% of patients were happy with their biosimilar. But 28% were worried they weren’t told they’d been switched. Transparency matters. That’s why states like Arizona require pharmacists to notify patients, record what was dispensed, and send a note to the doctor within five days. But not all states do that. And insurance companies? They often push for automatic substitution because it saves them money. A 2022 analysis found 78% of commercial plans require it - if the law allows.
Why This Matters for Your Health
The big promise of interchangeability? Lower costs. Biologics - drugs like Humira, Enbrel, or insulin - can cost $2,000 to $4,000 a month. Biosimilars typically run 15-30% less. That’s huge for people on high-deductible plans or without good insurance. The RAND Corporation estimated that if biosimilars hit their full potential, they could save the U.S. system over $100 billion over ten years.But there’s a flip side. A 2021 study in JAMA Dermatology found that when patients with psoriasis were switched from Humira to a biosimilar without medical reason, 20.3% stopped treatment entirely. Why? Maybe they felt uneasy. Maybe they had a flare-up. Maybe they didn’t trust the change. That’s why some doctors - like Dr. Kevin Winthrop from Oregon Health & Science University - argue that automatic substitution might hurt long-term care. If a patient loses trust, they stop taking their medicine. That’s worse than paying more.

What’s Coming Next?
The debate is heating up. In 2022, a bill called the Biosimilar Red Tape Elimination Act was introduced. It wanted to scrap the switching studies entirely and make every FDA-approved biosimilar automatically interchangeable. Supporters say it’s long overdue - why make companies do extra studies if all biosimilars are already proven safe? Opponents, including big drugmakers, say it cuts corners. The FDA is already moving toward streamlining the process. In 2023, they released draft guidance to simplify the switching study requirements.By 2026, Evaluate Pharma predicts biosimilars will capture 47% of the $168 billion biologics market. That’s billions in savings - if the system works. But right now, it’s a patchwork. Pharmacists are learning. Patients are confused. Doctors are cautious. And lawmakers are still figuring out how to balance cost, safety, and control.
What You Should Do
If you’re on a biologic:- Ask your doctor: "Is my drug eligible for a biosimilar?"
- Ask your pharmacist: "Am I getting the brand or a biosimilar?"
- Check your prescription label - it must list the manufacturer.
- If you’re switched without warning, call your doctor. Not all reactions are obvious.
- Know your state’s law. A quick Google search for "your state biosimilar substitution law" will tell you if substitution is automatic or requires approval.
There’s no one-size-fits-all answer. But being informed gives you power. You don’t have to accept a switch you’re uncomfortable with. You can ask for the original. You can ask for a biosimilar. You can ask for a reason. The system isn’t perfect - but you’re not powerless in it.
Can any biosimilar be automatically substituted?
No. Only biosimilars that have received an "interchangeable" designation from the FDA can be swapped automatically by pharmacists. As of late 2023, only 10 out of 41 approved biosimilars have this status. All biosimilars are safe and effective, but interchangeability is a separate legal and regulatory step focused on substitution rules, not quality.
How is interchangeable different from a generic drug?
Generic drugs are exact chemical copies of brand-name pills. They’re made in labs using simple processes. Biosimilars are copies of complex biologic drugs made from living cells - like proteins or antibodies. You can’t make an exact copy, so they’re "similar," not identical. Generics have been interchangeable for decades under the Hatch-Waxman Act. Biosimilars need extra studies - including multiple patient switches - to earn interchangeability status. That’s why only a few have it.
Can I be switched to a different biosimilar without my doctor’s approval?
No. Interchangeability only applies to swapping a biosimilar for the original reference product - not for switching between two different biosimilars. For example, if you’re on Cyltezo (an interchangeable biosimilar for Humira), your pharmacist can’t swap you to Hyrimoz (another biosimilar for Humira) without a new prescription. The FDA doesn’t allow that. Each biosimilar must be approved individually for interchangeability with the original, not with each other.
Do all states allow automatic substitution?
No. As of 2023, 40 states and Washington D.C. allow pharmacists to substitute interchangeable biosimilars without contacting the prescriber. But four states - Alabama, Indiana, South Carolina, and Washington - require prescriber approval. Six states and D.C. only allow substitution if it lowers the patient’s cost. This patchwork creates confusion for pharmacies and patients who travel or get insurance from out-of-state.
What should I do if I’m switched to a biosimilar without knowing?
First, check the label on your prescription bottle - it must list the manufacturer and product name. If you notice a change and weren’t told, call your pharmacist and ask why. Then contact your doctor. Some states require pharmacists to notify patients and send a note to the prescriber. If you feel worse, have new side effects, or your condition isn’t controlled, don’t ignore it. Ask for your original medication. You have the right to refuse a substitution, even if it’s allowed by law.


Kal Lambert
18 March / 2026Interchangeable biosimilars are a win for patients who can't afford $4k/month insulin. The science is solid. Let pharmacists do their job.
Stop overcomplicating it.